What’s Wrong with Medicine Today: My Perspective After Forty Years in the System
In Science We Trust | Part Three
OP
As physicians we prescribe a medication with the understanding that decision reflects the accumulated weight of scientific evidence. But what happens when science itself becomes the business. Who are we left to trust then?
Who Funds the Evidence
The entity with the greatest financial interest in the outcome of clinical research is also its primary funder.
Let that fact sink in for a moment.
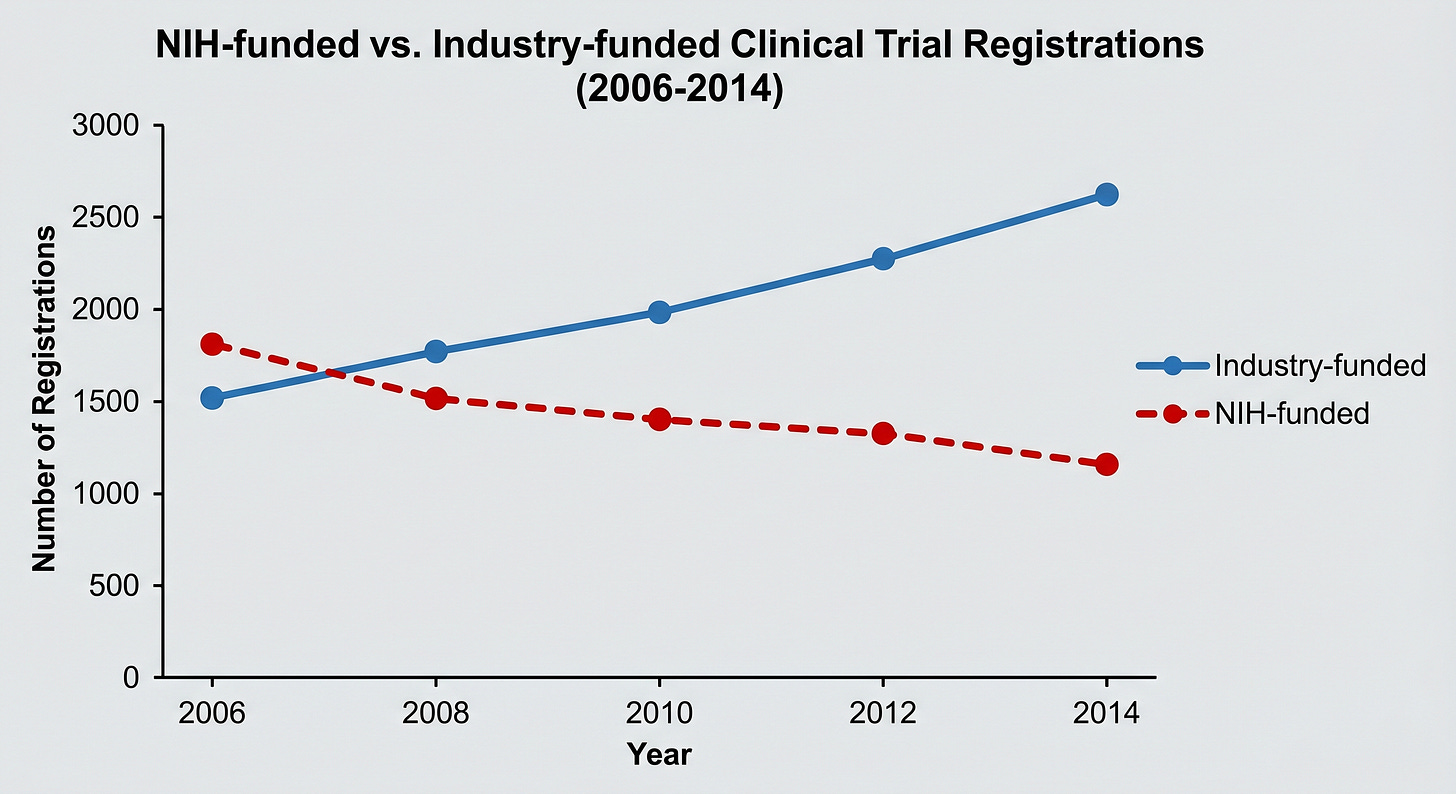
Between 2006 and 2014, the number of NIH-funded clinical trials registered at ClinicalTrials.gov fell 24 percent, from 1,376 to 1,048, while industry-funded trials grew 43 percent, from 4,585 to 6,550 (Ehrhardt et al., JAMA, 2015). The NIH budget fell 14 percent in real terms over the same period, which explains part of the decline.
How Outcomes Get Shaped Without Falsifying Data
Industry-funded trials produce more favorable outcomes for sponsored products than independently funded research. It has been replicated across multiple independent reviews over two decades and published in the highest-tier medical literature (Bekelman et al., JAMA, 2003; Lexchin et al., BMJ, 2003; Lundh et al., Cochrane Database of Systematic Reviews, 2017).
Trial design can be manipulated to favor a sponsor’s product before a single patient is enrolled:
Selecting patient populations where the drug performs best, choosing dosing regimens that favor the treatment arm, selecting a comparator at sub-therapeutic dose so the experimental drug appears superior.
Composite endpoints combine a hard outcome like death with softer outcomes like hospitalization, inflating the apparent magnitude of benefit.
Selective publication ensures that positive trials appear in the literature promptly while negative trials are delayed or never published, producing a body of evidence that systematically overstates benefit.
Truncated follow-up ends a trial before long-term harms have time to accumulate.
Subgroup mining searches through disaggregated data until a favorable result surfaces, then publishes that result as if it were the primary finding.
Technical accuracy and intellectual honesty are not the same thing, and a system engineered to produce misleading conclusions from accurate data is a dishonest system.
How Truthful Numbers Lie
The ASCOT-LLA trial, published in The Lancet in 2003, found that atorvastatin reduced the risk of heart attack by 36 percent in the studied population (Sever et al., Lancet, 2003). That figure, the relative risk reduction, was mathematically accurate and appeared in direct-to-consumer advertising.
What the advertising did not communicate was the absolute risk reduction: the heart attack rate fell from approximately 3.0 percent in the placebo group to 1.9 percent in the treatment group over the study period. The 36 percent relative risk reduction is simply the mathematical expression of that 1.1 percentage point absolute difference as a proportion of the 3.0 percent baseline risk.
Both numbers describe the same finding. They produce entirely different impressions of benefit in the mind of a patient or a physician who is not trained to recognize which one is being presented.
Atorvastatin has demonstrated genuine clinical benefit in appropriate patient populations, and nothing in this example should be taken as stating otherwise. This practice is not unique to this drug or this campaign. It was and remains a pervasive feature of pharmaceutical marketing and of how trial results are framed in publications and communicated to the clinical community.
Statistically Significant, But Clinically Significant?
It appears in pharmaceutical marketing, journal abstracts, prescribing guidelines, and conversations between physicians and patients. It carries the authority of clinical judgment implying that someone with medical training has evaluated and determined it matters.
A trial with enough participants can demonstrate statistical significance for an effect so small it changes nothing about how a patient feels or functions. The logical question becomes, is the effect clinically meaningful?: significant enough to matter in terms of how a patient feels, functions, survives, or dies. The final question is, do the benefit has clinical value?: whether the demonstrated effect is worth the cost or the risk, versus available alternatives.
The influenza antiviral oseltamivir (Tamiflu) is the clearest documented illustration of the gap between these three questions. The drug received FDA approval in 1999, promoted as reducing the severity and duration of influenza and preventing serious complications. Governments worldwide stockpiled it at a cost of billions of dollars during pandemic preparedness planning. The supporting evidence was largely manufacturer-funded, and the full underlying trial data was withheld from independent researchers for years.
When the Cochrane Collaboration obtained and analyzed the complete clinical study reports, the findings were precise (Jefferson et al., Cochrane Database of Systematic Reviews, 2014). Tamiflu reduced time to first alleviation of symptoms in otherwise healthy adults by 16.8 hours, from approximately seven days to 6.3 days. The independent also review found no good evidence to support reduction in hospitalizations, serious complications, or transmission.
Statistically significant, yes. Clinically meaningful or valuable, not in my opinion.
The Ghost-Writing Scandal
Merck created a systematic editorial process for its arthritis drug rofecoxib (Vioxx) in which company employees and contracted medical publishing companies wrote the manuscripts, determined the target journals, and arranged for the names of external academic investigators to appear as primary authors after the content was completed. Investigators received honoraria for lending their names to research they did not conduct and papers they did not write (Ross et al., JAMA, 2008).
This was documented through internal company materials released during litigation and subsequently published in JAMA. The drug was voluntarily withdrawn from the market in September 2004 after research demonstrated it doubled the risk of heart attack and stroke if taken for more than 18 months. It had been on the market for five years. Merck subsequently settled with plaintiffs for $4.85 billion.
On reviewing the JAMA documentation, the journal’s editor-in-chief, Catherine D. DeAngelis, stated: “The manipulation is disgusting. I just didn’t realize the extent.”. The Vioxx case don’t represent an isolated failure by a single company acting in an unusual way. The JAMA editorial accompanying the Ross findings noted explicitly that these practices were not uncommon, calling for immediate reform.
The FDA’s Structural Vulnerability
The FDA’s approval process requires the manufacturer to submit the clinical trial data supporting safety and efficacy. The agency evaluates that data but does not independently conduct or replicate the trials. The integrity of the process therefore depends entirely on the integrity of data submitted by the entity with the most direct financial interest in the approval.
Since its establishment in 1992, the accelerated approval pathway has allowed drugs onto the market based on surrogate endpoints, indirect biological measures assumed to predict clinical benefit, before actual clinical benefit has been demonstrated. Post-approval confirmatory trials are required to verify that benefit but in practice, nearly half (112 of 253) of the drugs authorized through this pathway had not been confirmed as clinically effective as of a 2021 BMJ investigation and only 16 had ever been withdrawn (Mahase, BMJ, 2021). Twenty-four of those unconfirmed drugs had been on the market for more than five years, some for more than two decades.
The Institute for Clinical and Economic Review reported in April 2021 that FDA’s reluctance to follow through on withdrawal when confirmatory trials were not completed meant that sponsors had little practical incentive to conduct them. Aduhelm — an Alzheimer’s drug approved at $56,000 per year despite internal FDA scientific objections, subsequently discontinued after confirmatory trials failed — is the most publicly visible recent illustration of what that failure costs. More than 30 oncology drug indications have been withdrawn in the past five years as the FDA recalibrates its standards.
Guideline Committees and Threshold Creep
Clinical practice guidelines are the mechanism by which research findings become prescribing behavior at scale. They tell physicians what to treat, when to begin, and with what. The committees that write them have historically included members with significant financial relationships with the pharmaceutical industry (Bekelman et al., JAMA, 2003; Angell, The Truth About the Drug Companies, 2004).
The documented pattern across cardiovascular medicine, lipid management, and diabetes care is consistent: treatment thresholds have been progressively lowered over time, more patients defined as requiring pharmacological intervention, and combination therapy established as standard of care with earlier initiation. Abramson and Wright documented concerns about this directional pattern specifically in lipid-lowering guidelines as far back as 2007, noting that the guidelines had moved ahead of the evidence base supporting them (Lancet, 2007).
What This Produces at the Bedside
The clinical endpoint of these intersecting forces is visible in the care of older patients across American medicine. Polypharmacy defined as five or more concurrent medications, is common and consequences well documented: increased adverse drug events, drug-drug interactions, falls, cognitive impairment, and hospitalizations (Masnoon et al., BMC Geriatrics, 2017; Hajjar et al., American Journal of Geriatric Pharmacotherapy, 2007).
I observed this repeatedly: elderly patients presenting with altered mentation, cognitive changes, confusion, behavioral disturbance, that on careful medication review proved to be the consequence of multiple centrally acting and psychoactive drugs prescribed by different providers, each subsequent prescription treating a symptom that was likely a side effect of from a prior one.
To add insult to injury, these patients stayed in the hospital, underwent a battery of additional tests despite the obvious cause, adding tens of thousand of dollars in unnecessary healthcare expenses.
A Reasonable Proposal and an Honest Assessment of Its Prospects
One simple structural reform could address this conflict: pharmaceutical companies seeking FDA approval should be required to contribute to a pooled, independently administered research fund from which grants are awarded to unaffiliated investigators who design and conduct the confirmatory trials the FDA requires. The company that developed the drug no longer has any control over the trial that evaluates it.
The reason of why it does not exist: the financial interests that would fund this arrangement are the same interests that fund the political process required to mandate it.
In Science We Still Trust… But…
My point here is not that medications in common use are not working or are unsafe. The point is we should all demand more transparency about conflict of interest in medical research and support independent research at a governmental level.
When a new prescription is proposed, three questions are worth asking:
What is the absolute risk reduction this drug provides for someone with my specific profile, not just the relative risk reduction?
What are the alternatives, including non-pharmacological ones?
If I am already taking multiple medications, who is reviewing the complete list for interactions?
These are not adversarial questions. They are the questions a well-informed patient asks. Your physician will recognize them as exactly that.
Found this article useful? Share your thoughts. Join the conversation below.
Educational content on The Metabolic Archives is free, because medical information should be accessible to everyone. If you find value and want to support the work, a paid subscription is available and genuinely appreciated. Visit the About Page for additional information.
The Metabolic Archives is for educational and informational purposes only, and is not intended as medical advice, diagnosis, or treatment, and does not constitute a doctor-patient relationship. Do not adopt any recommendation discussed in any article or guides published here, make changes or abandon any prescribed medical treatment without prior consultation with your physician. Always seek the advice of your physician or other qualified health provider for any questions regarding your medical condition and recommended treatment options.
By reading this post, you acknowledge that you have read and agree to the Terms of Service of The Metabolic Archives, which govern all use of this content including restrictions on reproduction.
© 2026 The Metabolic Archives. All rights reserved.